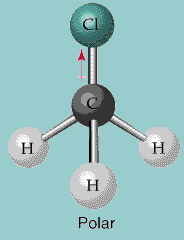
Each of these atoms is bonded to the central carbon atom, forming a tetrahedral shape. In CH2Cl2, the carbon atom is bonded to two chlorine atoms and two hydrogen atoms. The molecular geometry of CH2Cl2 can be determined by analyzing the arrangement of atoms and lone pairs around the central carbon atom. Determination of the shape of CH2CL2 molecule This means that the molecule has a positive and negative end, and it can interact with other polar molecules through dipole-dipole interactions. The asymmetrical distribution of electrons in the molecule makes it polar. As a result, the molecule has a dipole moment, with a partial negative charge on the chlorine atoms and a partial positive charge on the hydrogen atoms. In CH2Cl2, the carbon atom is bonded to two chlorine atoms and two hydrogen atoms, resulting in a tetrahedral shape with a dipole moment.Ĭhlorine has a higher electronegativity than carbon and hydrogen, which means that the electrons in the covalent bonds are pulled more towards the chlorine atoms. The polar/non-polar nature of the CH2Cl2 molecule can be explained by analyzing the electronegativity difference between the atoms in the molecule. Explanation of the polar/non-polar nature of CH2CL2 molecule Calculate the formal charges of each atom in the molecule to confirm the accuracy of the Lewis structure.Ĭ.In CH2Cl2, each atom has a complete octet or duet. Check that all atoms have a complete octet of valence electrons, except for hydrogen, which can only have two.In CH2Cl2, there are two remaining valence electrons, which are placed as a lone pair on the central carbon atom. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
